Linking laboratory testing to patient outcomes
Maximising the value of laboratory medicine
Mike Hallworth, Consultant Clinical Biochemist, Shrewsbury, UK
Clinical laboratory workers believe that the work they perform in providing laboratory tests is valuable. However, data to validate this has been limited, and evidence of the contribution of laboratory medicine to the overall process of diagnosis and management is not easy to obtain. Many articles and presentations seeking to promote the value of laboratory medicine have made use of what has become known as the “70 per cent claim”. This is presented in various forms, most commonly that “Laboratory Medicine influences 70 per cent of clinical decisions”, or minor variations around this figure. However, the data on which this estimate was based represents unpublished studies and anecdotal observations and cannot now be objectively verified. In addition, much of the evidence relating to the value of laboratory medicine is poorly structured and does not relate to clinical outcomes.
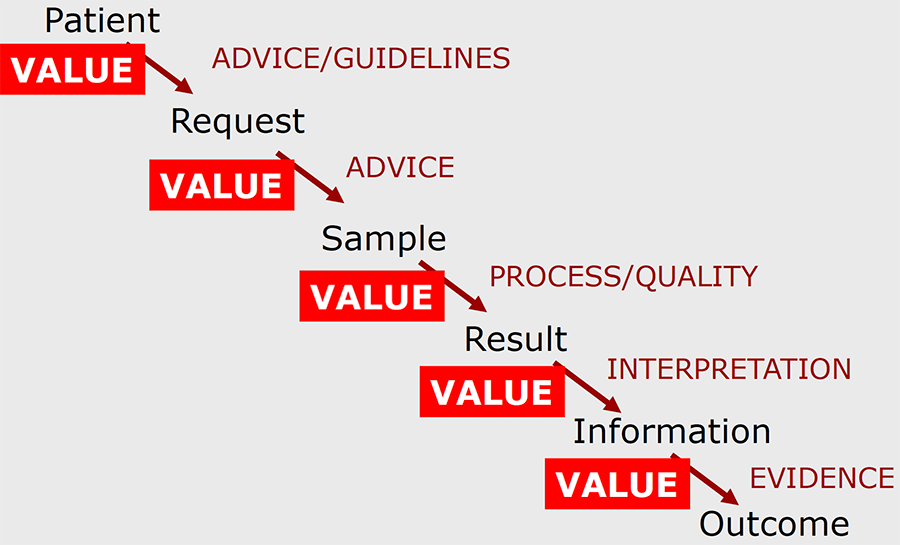
We need more specific and evidence-based measures of the added value of laboratory medicine, which in turn require better-designed studies and better use of existing biomarkers. The mindset within laboratories in the past has too often been that our starting point is a specimen (of blood, other body fluid, tissue or whatever) and our endpoint is an accurate result. This tends to lead to a ‘factory’ mentality for laboratory workers, such that all their attention is focussed on improving processes within the laboratory. Undoubtedly, this is important – but we need to remember that the true starting point is a patient, and the true endpoint is an improved clinical outcome for that patient (Figure 1). The laboratory can add value to the testing process at every point along the pathway, by advising on appropriate requesting and proper sample collection, ensuring efficient and high-quality analysis and then converting results into useful information, which can inform decisions about diagnosis and treatment.
Improved clinical outcomes
The definition of value in healthcare is normally taken as the ratio of outcome to cost. Thus, better value is obtained when improved clinical outcomes are obtained at a lower cost, or greatly improved outcomes are obtained with relatively lower increases in cost. We know that laboratory medicine is particularly well leveraged in that respect, and laboratory testing has been called the most cost-effective intervention in medicine.
Assessing the value of laboratory medicine, therefore, requires a close focus on the impact of testing protocols on outcomes, which in turn requires a standardised set of measures of clinical outcome in all relevant clinical conditions – a “common currency” for outcomes, which enables clinical laboratory researchers to communicate the effect of using new biomarkers in terms of outcomes that are familiar to clinicians.
Such standardised outcome measures have been absent in the past, but the work of the International Consortium for Health Outcomes Measurement (ICHOM) in bringing together international teams of patients, physicians and researchers to define outcomes that matter most to patients who live with different conditions is transforming this field and will allow much better analysis of the true clinical value of new biomarkers in specific situations.
A report by the Lewin Group in 2009 on the value of laboratory screening and diagnostic tests for prevention and healthcare improvement concluded that: “in order to improve outcomes, a laboratory test must be appropriately ordered, conducted, returned with results on a timely basis and affect a decision for further diagnosis and treatment”.
The work of Plebani’s group has clearly shown that where diagnostic error arises from laboratory testing, the pre- and post-analytical phases are much more vulnerable to error than the actual analytical phase, which implies that laboratories need to refocus their efforts on error reduction toward the total testing process rather than simply on the analytical aspects of their work.
Figure 2 shows an analysis of laboratory-related causes of diagnostic error. The first two causes – ordering of inappropriate tests and not ordering appropriate tests – are conveniently discussed together as better laboratory utilisation. This means working with clinicians to produce evidence-based guidelines on testing strategies, reducing inappropriate testing, which can lead to overdiagnosis and misdiagnosis, but – crucially – also ensuring that the right tests are done at the right point in the patient pathway, to ensure that the correct diagnosis is reached at the earliest possible point. As tests become more complex (particularly in the area of molecular genetics), specialist advice from the laboratory is vital to ensure that test selection is appropriate.
The third area listed in Figure 2, which can contribute to diagnostic error is a failure to use an appropriate test result properly, due to lack of understanding of the significance of the result or failure to integrate the test result into the overall clinical picture and the results of other investigations. The U.S. Institute of Medicine, in their 2015 report “Improving Diagnosis in Health Care” recommended that health care organisations should ensure that healthcare professionals should have the appropriate knowledge, skills, resources and support to engage in the diagnostic process, and laboratory workers have a crucial role in providing education in the proper use of laboratory testing and interpretation of results in specific cases.
Outcome-based evaluations
However good a laboratory test, it cannot affect the individual patient outcome if the result does not reach the clinician who is responsible for delivering care when it is needed (Figure 2, number 4). Test result management has been named as the #1 patient safety concern for 2019 by the U.S. ECRI Institute and urgent physician notification of critical results, both quantitative and qualitative, has become part of the standard of care because of the high impact it has on patient welfare and clinical outcomes. This means that laboratories must work with their users to decide, which results need to be communicated urgently, and by what means. It also means the development of effective electronic systems to indicate that a particular result has been seen and actioned by the relevant clinician, and (in some cases) follow-up by the laboratory of critical results, which have not been viewed or actioned.
The final point of Figure 2 – an appropriate test result which is wrong or inaccurate – is the traditional focus of laboratory quality management procedures. Constant vigilance in this area is essential, but (as mentioned above) it currently represents the least common cause of the diagnostic error.
In addition to efforts to improve how existing tests are used, it is vitally important that new biomarkers receive robust outcome-based evaluations before they are introduced into clinical practice so that the intended benefits of the biomarker are clearly understood, including the role of the new test, how and where it fits into the testing pathway (screening/triage/confirmation) and the clinical performance requirements for the test. The Test Evaluation Working Group of the European Federation of Clinical Chemistry and Laboratory Medicine have produced excellent work in this area including checklists for test evaluation
Laboratory doctors and scientists of the future must be involved in producing guidelines for investigation, advising clinical staff on the best strategy for individual clinical presentations and the further tests needed to confirm a diagnosis, and ensuring that results are not misinterpreted or missed and that resources (human, technical and financial) are used to do the right test on the right person at the right time. It’s a daunting challenge, but getting this right means better use of tests, better patient care, lower healthcare costs, improved job satisfaction for laboratory workers and enhanced ability to recruit and retain good scientists in laboratory medicine. That’s a goal worth working for!