Biosystems - New Fecal Occult Blood System
Colorectal cancer (CRC) is the third most common cancer and the fourth leading cause of cancer-related deaths worldwide. Fortunately CRC can be a treatable, beatable and preventable cancer, if is detected in early stages of the disease. The detection of fecal occult blood, is the key point to prevent colorectal cancer among population.
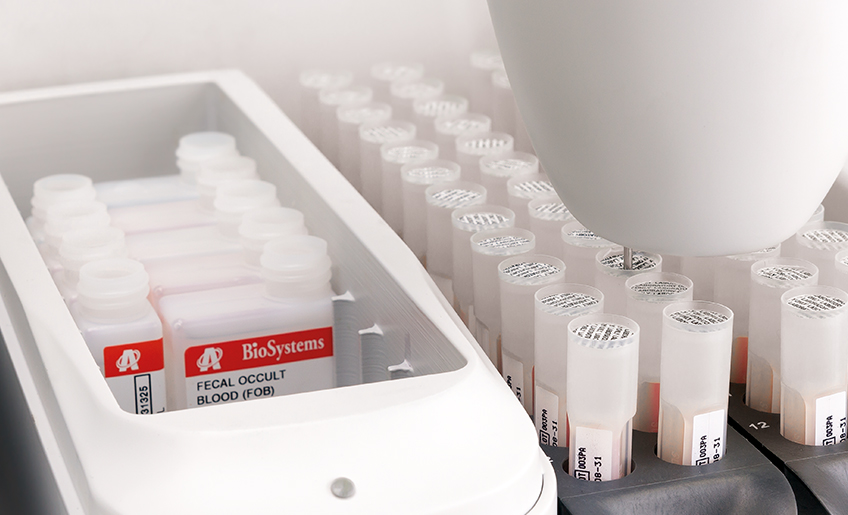
The new Fecal Occult Blood (iFOB) integral system, developed by Biosystems, provides the best diagnostic solution for FOB testing. The new Fecal Occult blood integral system is composed by an easy-to-use stool collection tube for easy sampling at patient’s home or in the laboratory, an improve immunoturbidimetric assay by means of a new and high sensitive antibody against human haemoglobin (no cross reaction with non-human Hb) and an update of our well-known A15 analyser with a new cap piercing function (no need to decap or recap samples tube after testing) and a dedicate software for fecal samples.
By using our integral system, we provide the most accurate, reliable and safe method for FOB testing, making this test accessible to any kind of laboratory.
In addition to FOB tests, our integral systems can be updated with new fecal tests such as fecal Calprotectin, Transferrin and Helicobacter pylori, making this system a complete solution for stool samples analysis in the laboratory.










































































