Improving Patient Care and Reducing Costs in the Laboratory by Optimising Test Utilisation
Article provided by the Clinical and Laboratory Standards Institute
Proper utilisation of laboratory testing is an important factor in improving patient care outcomes. Clinically useful laboratory tests provide timely diagnosis, identify at-risk patients in need of intervention, accurately predict prognosis, guide therapy, and effectively monitor treatment. In contrast, inappropriate laboratory test utilisation reduces the value of laboratory testing and may result in diagnostic errors or adverse patient care outcomes. While implementation of appropriate test utilisation practices in the laboratory is an important factor in improving patient care outcomes, it can also reduce costs.
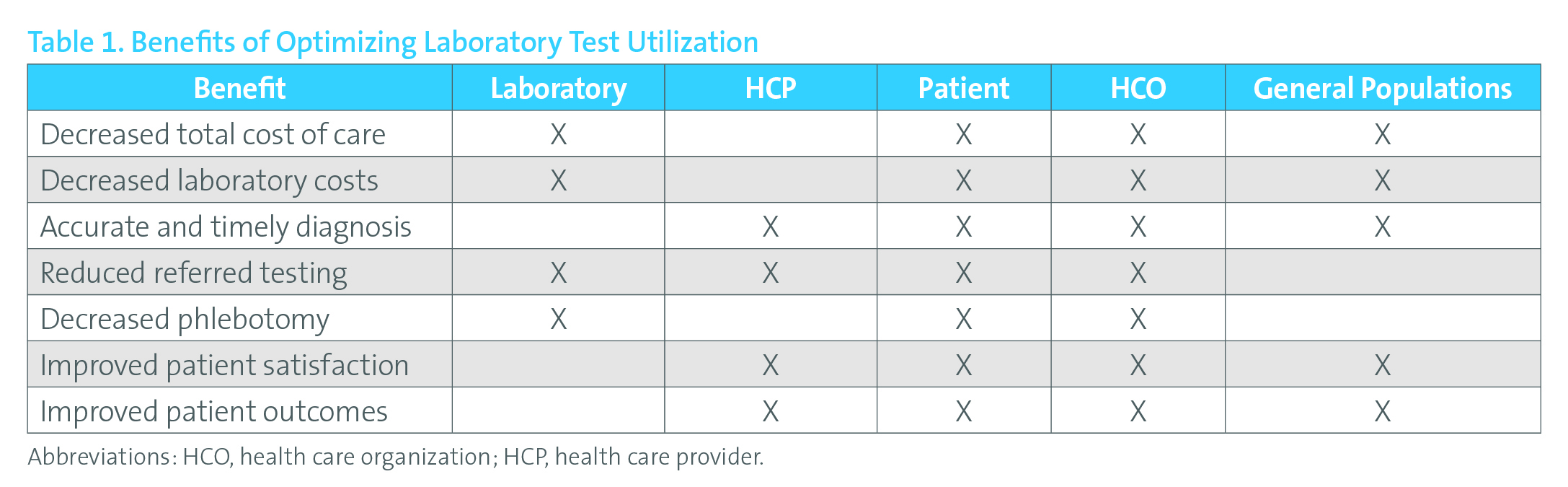
Table 1. Benefits of Optimising Laboratory Test Utilisation
Reprinted with permission from Clinical and Laboratory Standards Institute from: CLSI. Developing and Managing a Medical Laboratory (Test) Utilization Management Program. 1st ed. CLSI document GP49. Wayne, PA: Clinical and Laboratory Standards Institute; 2017.
Changes in healthcare reimbursement models are another reason laboratory test utilisation has gained interest. Highly specialised esoteric tests are more readily available and demand for their use has increased. The overutilisation of these and other laboratory tests contributes to high healthcare costs without adding value for the patient or the healthcare system.
The five main reasons for inappropriate laboratory testing are:
- Inappropriate test selection on the part of the clinician
- Incorrect test procedure performed by the laboratory
- Misinterpretation of results
- Omission of clinically appropriate testing (underutilisation)
- Failure of the healthcare provider to follow up on test results with the patient
An organisation can develop solutions for test utilisation improvement by understanding the reasons for inappropriate use of laboratory tests. Selecting the proper diagnostic test in a particular clinical situation has become more difficult as the number and complexity of tests expand. While clinician and patient education is helpful, alone it is insufficient to ensure optimal test utilisation.
What is Test Overutilisation?
Test “overutilisation” is the performance of a test for which the result has little to no effect on the patient’s care. There are many causes of test overutilisation, most of which originate during the pre-examination or pre-analytic phase of testing.
One of the causes of test overutilisation is that a significant portion of laboratory test orders are “redundant” or “duplicate,” in that the test is repeated too soon to provide useful information. There are several factors that contribute to duplicate testing. One is that large organisations with many healthcare providers face the challenge that one clinician may not be aware that a test was already ordered by another clinician. Which tests have already been ordered and are pending is not always readily available to the busy clinician. Electronic notifications and stopping unnecessary duplicate orders at the point of order entry is a proven way to decrease this type of overutilisation.
Another type of test overutilisation occurs when tests are ordered more frequently than necessary. Unnecessary, repeat testing typically involves routine, high-volume tests. Causes include standing orders, inpatient phlebotomy scheduling, or uncertainty about reasons for retesting.
Testing redundancy occurs when two or more tests ordered together are expected to provide the same information. Some tests provide essentially the same information and, when ordered at the same time, provide little if any additional information. In some cases, one test is preferred over another. Working with providers to develop and implement best-practice test algorithms can substantially reduce this type of redundancy.
Technical problems also contribute to testing overutilisation. Misorders are one type of technical problem, and may be caused by marking the wrong test(s) order forms or by the laboratory processing the order incorrectly. A second type of technical problem is that a test may be inappropriately selected to evaluate a medical condition for which it is not indicated. Pathologists and other laboratorians should work closely with their information technology personnel to remove obsolete tests from the test menu, and make it easy for providers to order the correct test and difficult to order the wrong, sound-alike test.
Test Underutilisation
The omission of clinically useful testing is one instance of test underutilisation. Working with providers to implement appropriate testing algorithms is an excellent way to work to assure necessary testing is performed at the appropriate time. These algorithms, particularly if automated, could prove very helpful for population health management. Some reasons for test underutilisation include incomplete initial or reflex testing, insufficient laboratory monitoring of a disorder or treatment, and lack of expected or recommended testing needed to assess a clinical condition. Similarly, best-practice algorithms that include appropriate reflex testing are helpful to ensure important tests are performed as necessary. Although it is not studied as often as overutilisation, underutilisation of laboratory tests may cause unfavourable clinical consequences. Addressing underutilisation holds great promise for disease prevention and the optimal management of chronic diseases.
Implementing a Test Utilisation Programme
It is important that the organisation’s medical and administrative leadership understands the importance of managing utilisation throughout the organisation. Leadership can show support for a test utilisation management programme by establishing and participating in the programme.
Participation from many areas of the healthcare organisation is essential to the proper implementation and success of a test utilisation programme. Multispeciality representation and participation is an important consideration in the formation of a team. The following individuals and/or groups add value to a laboratory utilisation programme:
- Pathologists and doctoral-level laboratory scientists
- Physicians, clinicians, and nurses
- Administrators, managers, and supervisors
- IT and data services
- Quality and/or continual improvement professionals
- Financial analysts and accountants
- Statisticians and public health experts
- Medical laboratory scientists
Utilisation committees help manage change and provide guidance on test utilisation. In some organisations, the utilisation committee reviews and approves requests for existing and new referred tests (eg., expensive genetic and molecular tests) submitted to referral laboratories. Others review the scope, use, and effectiveness (both cost and clinical) of current laboratory services, as well as any new laboratory services and associated technology requested by clinical stakeholders.
Although the advantages are significant, a formal committee structure is not always suitable for every organisation. Some organisations may institute a less formal working group or, depending on the resources and expertise available, smaller subcommittees and task forces made up of personnel with expertise appropriate to a specific utilisation issue.
Utilisation priorities vary based on the organisation’s size, the types of patients served, and the resources available for analysis. Many laboratories begin with projects involving higher-cost referral laboratory tests. The financial effect of the proper utilisation of these tests is relatively easy to track, because the costs are captured separately from the laboratory’s in-house testing costs.
Laboratory utilisation management programmes often focus their efforts on:
- Clinical and anatomic pathology services
- Referred testing
- Esoteric testing, especially genetic and molecular
- Point-of-care testing
- General laboratory quality improvement
- Sample suitability
- Turnaround times
- Frequency of testing, including duplicate testing
The Importance of Measuring Outcomes of a Test Utilisation Programme
Statistical and decision support analyses, as well as financial information (for actual testing and effects of test utilisation on other healthcare expenses or revenues), are valuable in assessing appropriate and inappropriate test utilisation and for monitoring the effectiveness of the programme. Information is available that suggests what to measure, how to measure selected metrics, and how to construct meaningful reports.
Conclusion
Implementation of a test utilisation programme is an effective way to reduce costs and improve patient outcomes. CLSI has recently published Developing and Managing a Medical Laboratory (Test) Utilization Management Program, 1st ed. (GP49), guidance for the initiation, development, and maintenance of an effective test utilisation programme in the laboratory.
References available on request.