Transformation Hub & Talks New
Future of laboratory medicine in the next decade
3-6 February 2020
Za'abeel hall 2, Dubai World Trade Centre
New at Medlab Middle East, the Transformation Hub will allow you to identify technology trends & products, address key questions and provide solutions to implementing advanced technology in the laboratory setting.
It brings business executives and healthcare professionals to explore innovative actions in transforming diagnostic services to deliver quality and rapid tests.
As an add on feature, future lab professionals will also hear from lab champions and gurus on various career opportunities in the laboratory medicine industry, in the special sessions on Career Talks.
Also launching is the new Transformation Hub, this area next to the talks gives you the chance to see and experience state-of-the-art laboratory solutions.
Visit Za'abeel hall 2 to experience the future of diagnostics...
Why the Transformation Hub?
Key topics:
- Technology acceleration
- Improving care with automation
- Artificial Intelligence
- Digital health
- NGS
- Genetics
- Genomics
Benefits of attending:
- Discover new and upcoming trends in laboratory management
- Discover application of hi-tech solutions in laboratory setting
- Review showcase of products and services and their applications
- Investigate the role of technology in improving patient engagement and health awareness
- Assess how disruptive technologies and innovations can impact the laboratory
- Determine how connected health technologies can support lab testing and analysis
Featured products
Spartan CYP2C19 Test
CYP2C19 is a liver enzyme that metabolizes 15% of all prescribed drugs, including anti-platelet drugs, antidepressants, and PPIs. Carrying a CYP2C19 mutation can impair drug metabolism. In fact, 30% of Caucasians and 50% of Asians and Indians carry CYP2C19 mutations that impair drug metabolism.
Spartan ApoE Test
There is a market need for a rapid and inexpensive screening test that can be used to easily identify patients carrying the E4/E4 genotype. These patients are at a higher genetic risk for developing Alzheimer’s Disease (AD), and may also respond differently to various AD therapeutics. The Spartan ApoE System is designed to meet this need by providing a rapid, on-site solution for patient screening.
QIAstat-Dx
The next generation of syndromic insights
Utilizing real-time PCR, the QIAstat-Dx Analyzer reports on comprehensive panels for respiratory and gastrointestinal targets in about an hour. QIAstat-Dx is the only instrument on the market with both bidirectional LIS connectivity and the ability to export Ct values and endpoint fluorescence values for targets.
The Respiratory Panel analyzes 21 viral and bacterial pathogens, to differentiate the causes of acute respiratory tract infections.
The Gastrointestinal Panel includes comprehensive coverage detecting the 24 most common viral, bacterial and parasitic targets that cause GI infections.
The Meningitis/Encephalitis Panel is currently under development to provide clinical insights for patients with suspected meningitis or encephalitis infections.
PRC|ONE
The PCR|ONE system is a fully automated, multiplexed PCR system for sample preparation, amplification, and detection of DNA in 15 minutes—a true “sample-in-answer out” technology for rapid point-of-care diagnostics of infections.
The PCR|ONE system is the first completely automated, multiplexed, gold standard PCR system with 15-minute turnaround. The use of physical multiplexing makes the system ready for rapid broadening or modification of the panel of genes inspected, making it responsive to infections as they spread and evolve. This system will be disruptive in the fight against HAIs because it accelerates and simplifies comprehensive diagnosis, enabling comprehensive molecular diagnosis in the admission ward for the first time, reducing the risk of infections, and saving costs and time associated with pathogen isolation.
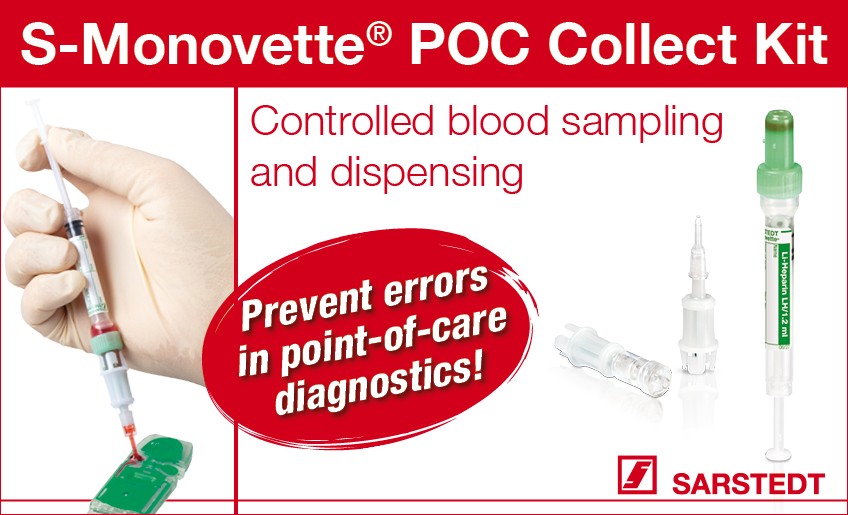
S-Monovette® POC
Collect Kit
The S-Monovette® POC Collect Kit, test cartridge sample dosing can be carried out accurately and in just one step. This provides reliable results quickly, while ensuring optimal patient care and turnaround times. It reduces the risk of infection for the user by ensuring hygienic dispensing.
Advantages for users and patients
- Rapid therapy decisions thanks to quick turnaround time
- Risk of infection minimized thanks to closed blood transfer
- Needle-free POCT DISPENSER transfer unit
Cost and quality advantages
- Reliable results thanks to more precise sample dosing
- Reduction in consumables to disposed of
- Reduced process costs